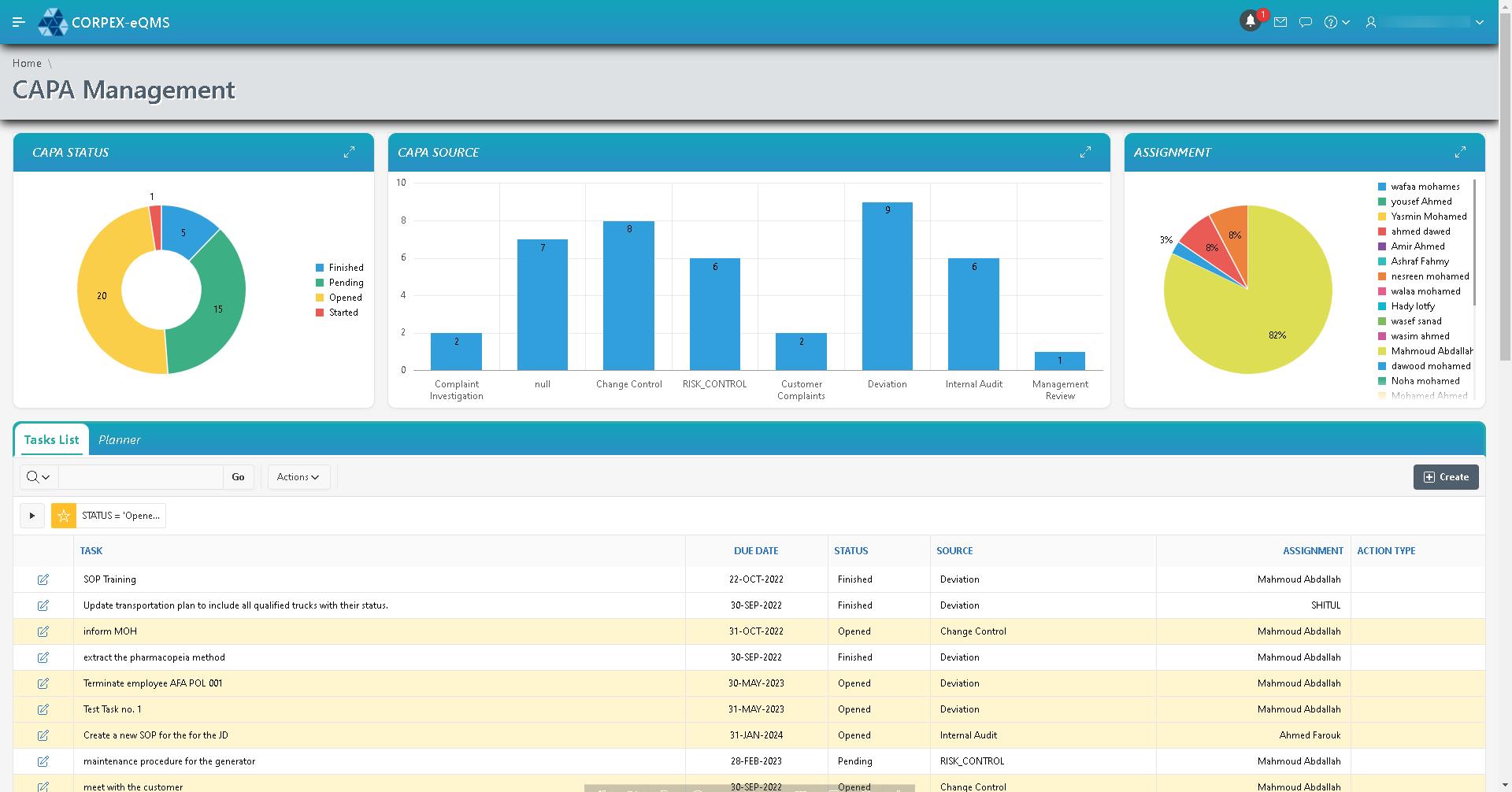
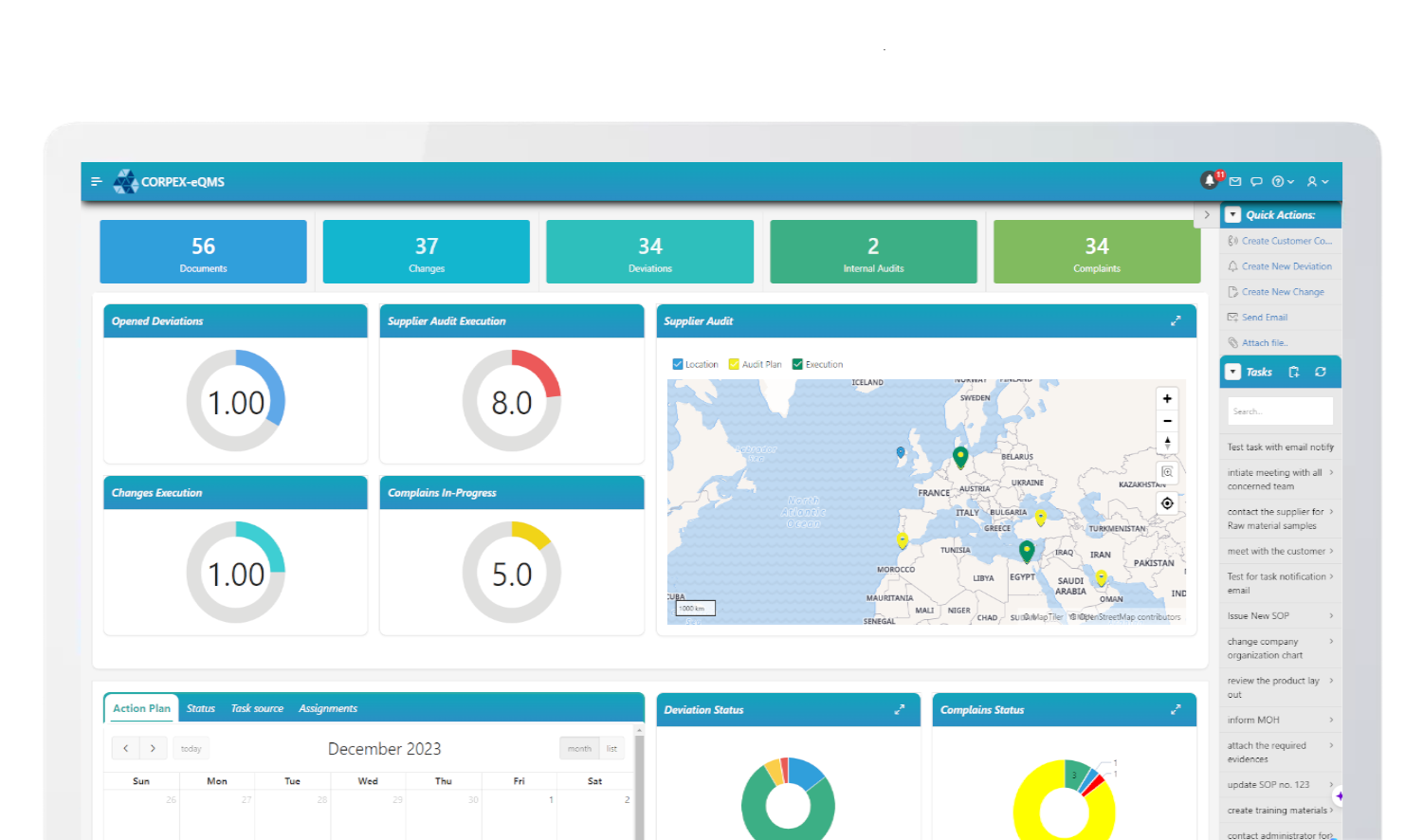
CAPA Management
Streamlining Your Path to Quality:
In the relentless pursuit of excellence, every organization encounters deviations from optimal performance. These departures, while inevitable, present valuable opportunities for growth and improvement. This is where Corrective and Preventive Action (CAPA) Management comes into play, serving as the compass that guides your Quality Management System (QMS) towards continuous advancement. And, within the robust framework of CORPEX-QMS, the CORPEX-CAPA module emerges as your indispensable partner in this critical endeavor.



