Cleantrol EM™
Innovative Solution for Viable Monitoring of Controlled Environments.
Interactive Environmental Control
Cleantrol is a risk-based environmental monitoring solution that manages sampling of controlled environments for viable air, viable particulates, and surface viables. It allows for the assessment of cleaning and disinfection effectiveness, identification of trends, and early detection of potential problems.
Cleantrol manages and automates EM data through a daily interactive calendar and workload manager to facilitate EM automation, simultaneous qualification of process capabilities, and integral handling of deviations.
"The FDA believes manual systems of environmental monitoring (EM) have large inherent operational risks... Computer trending for pattern recognition is the most efficient and effective way to handle such large quantities of data."
Key Core Features
Comprehensive tools for total environmental control.
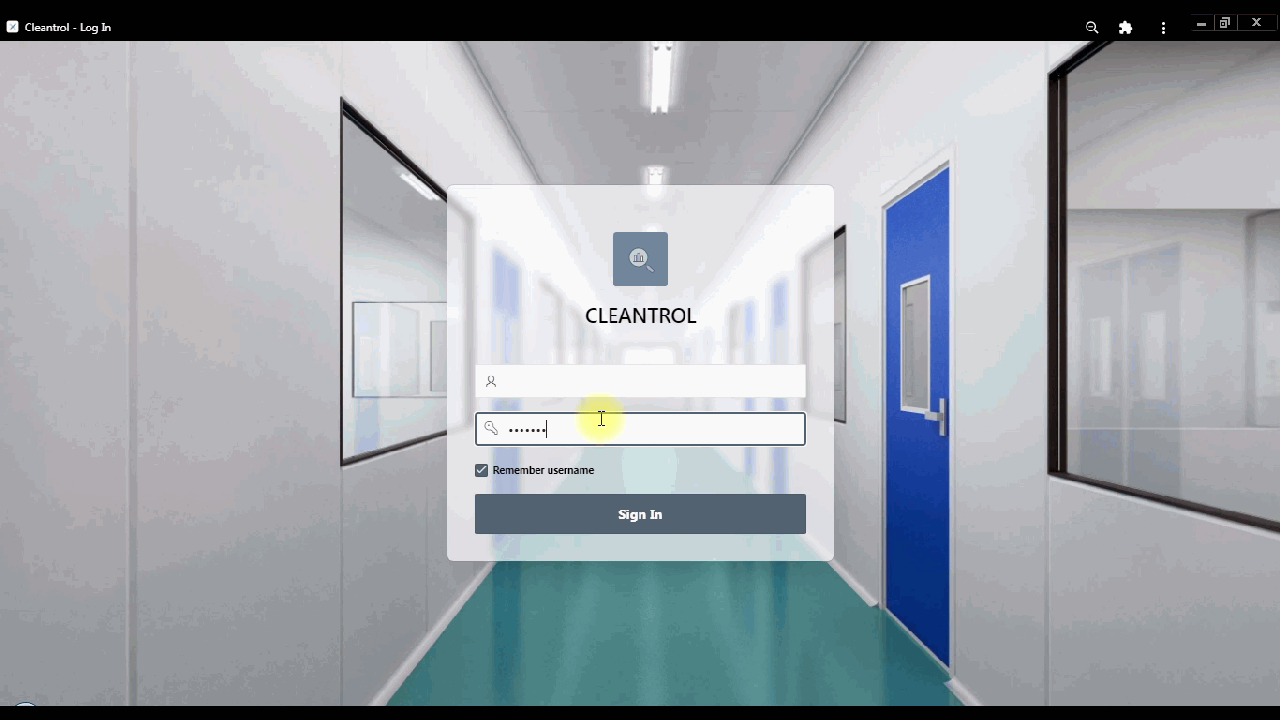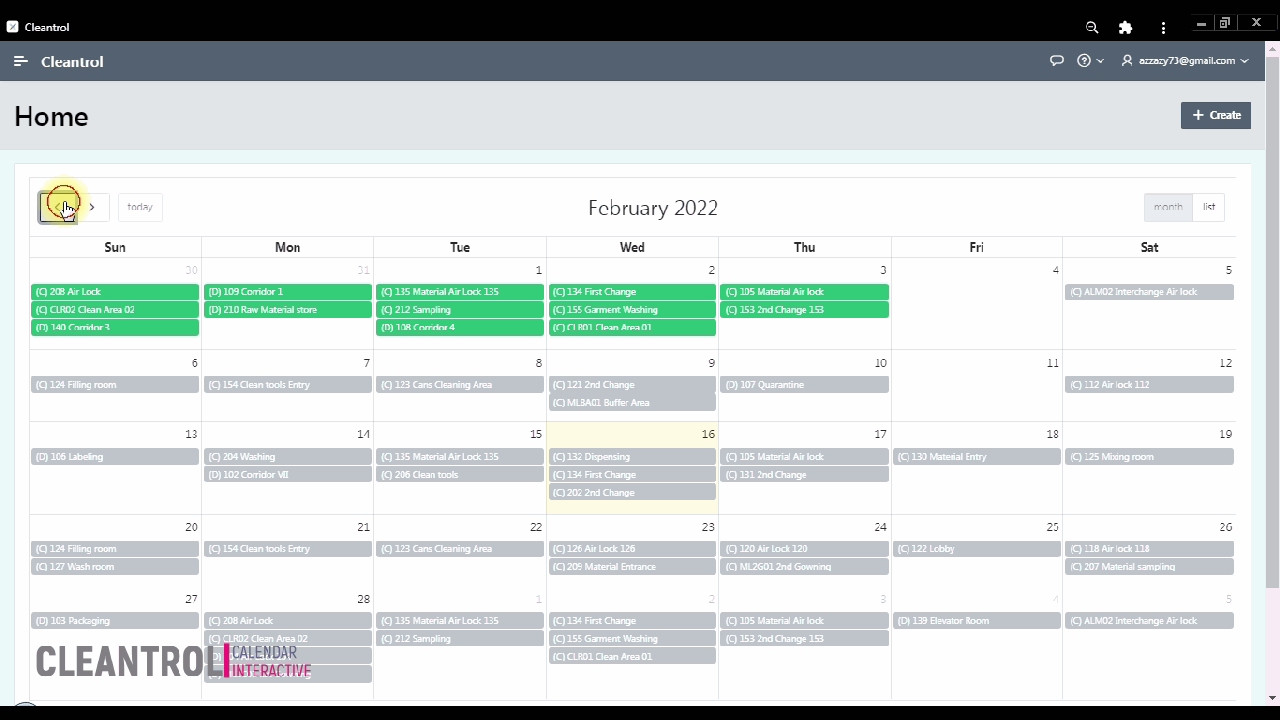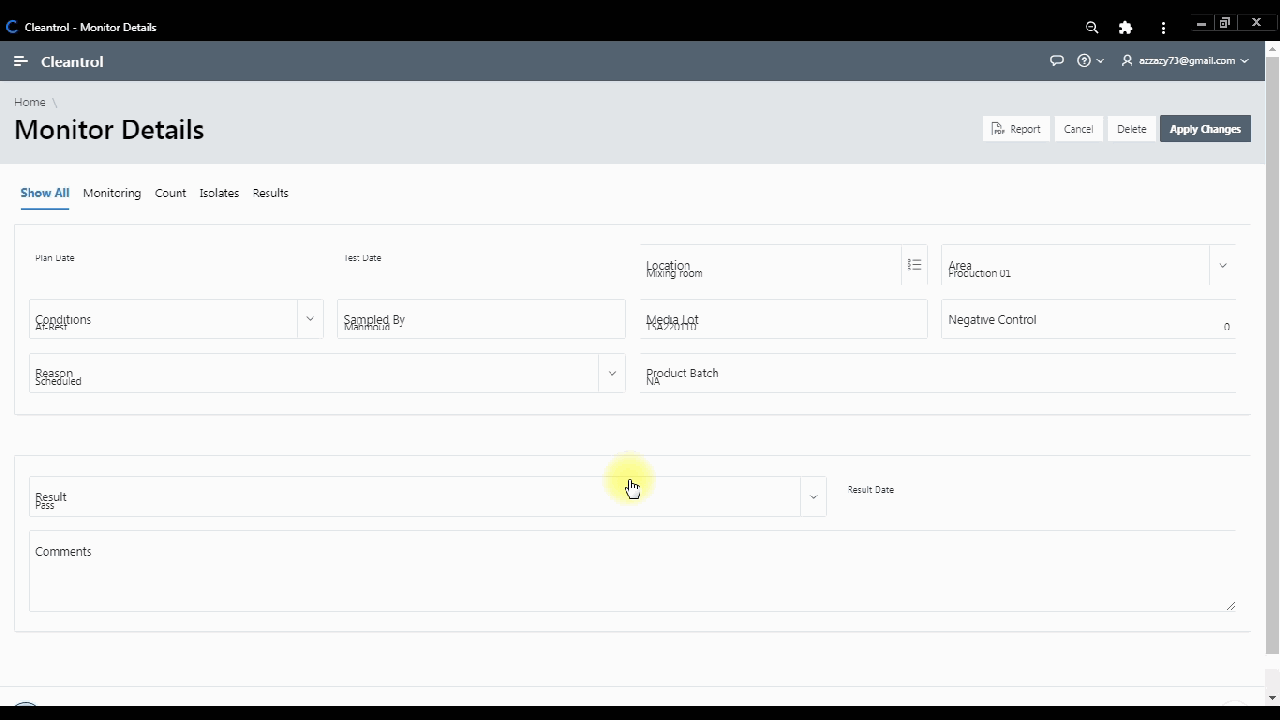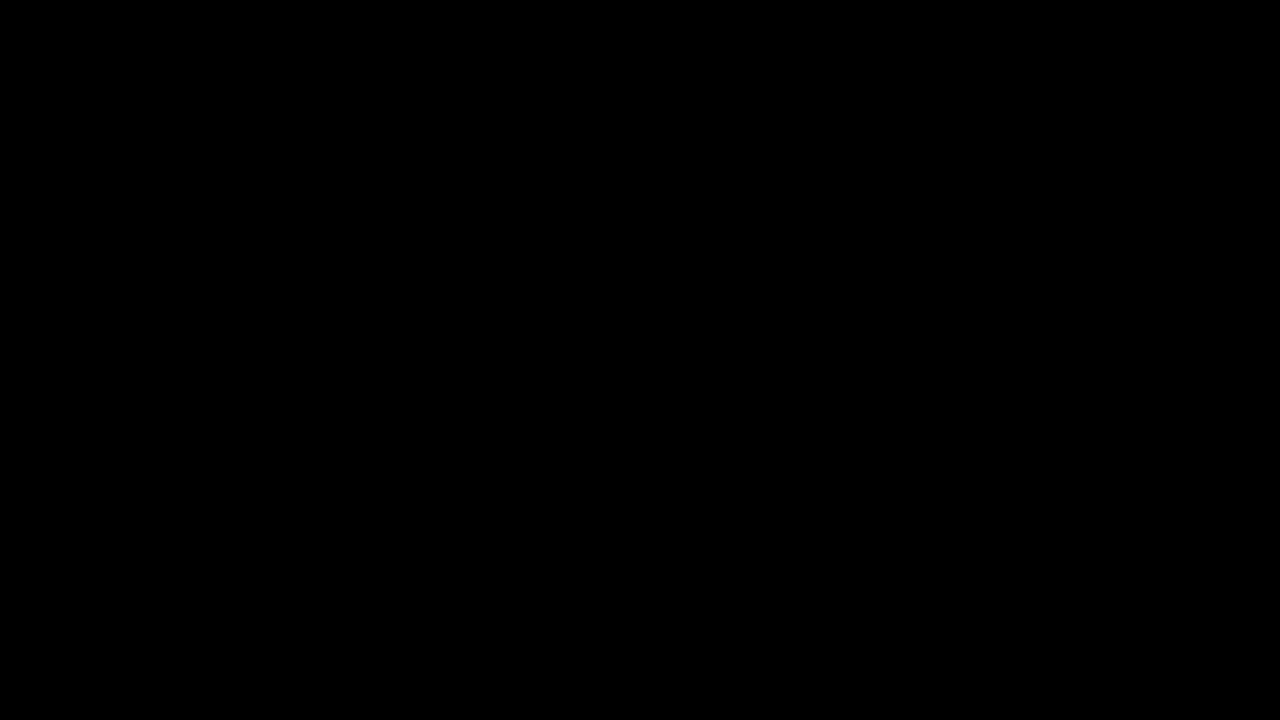
Interactive Schedule
Monthly interactive calendar arranges day-to-day monitoring tasks. Access monitoring details directly to track viable counts and organize activities perfectly.
Instant Reporting
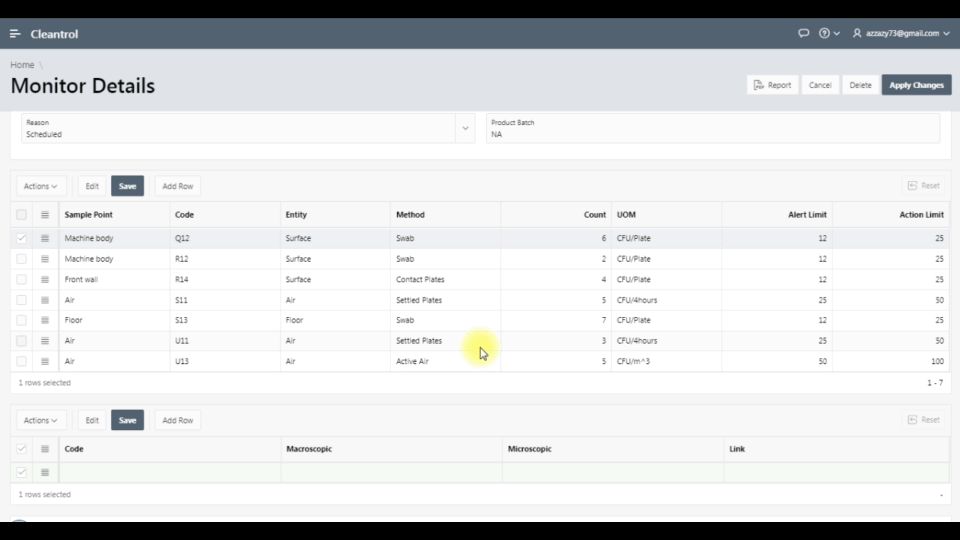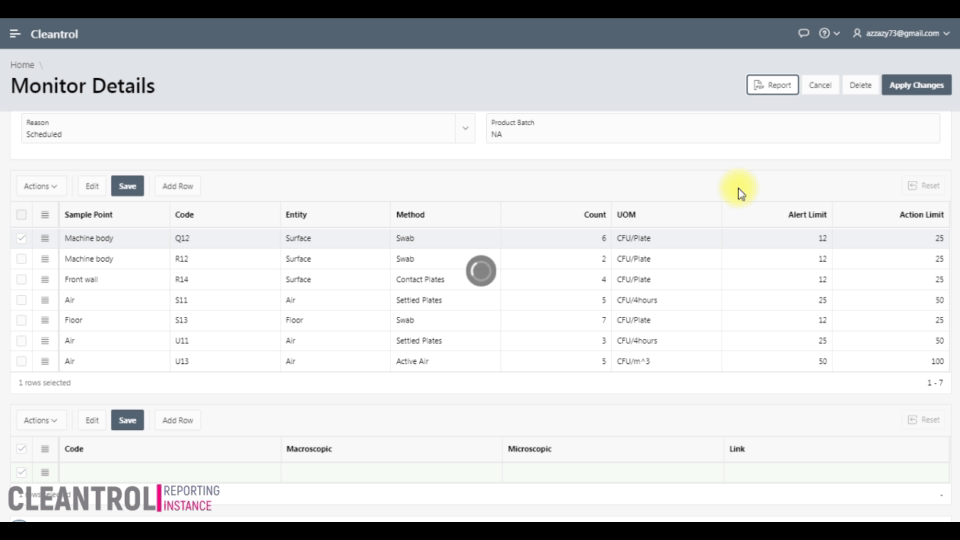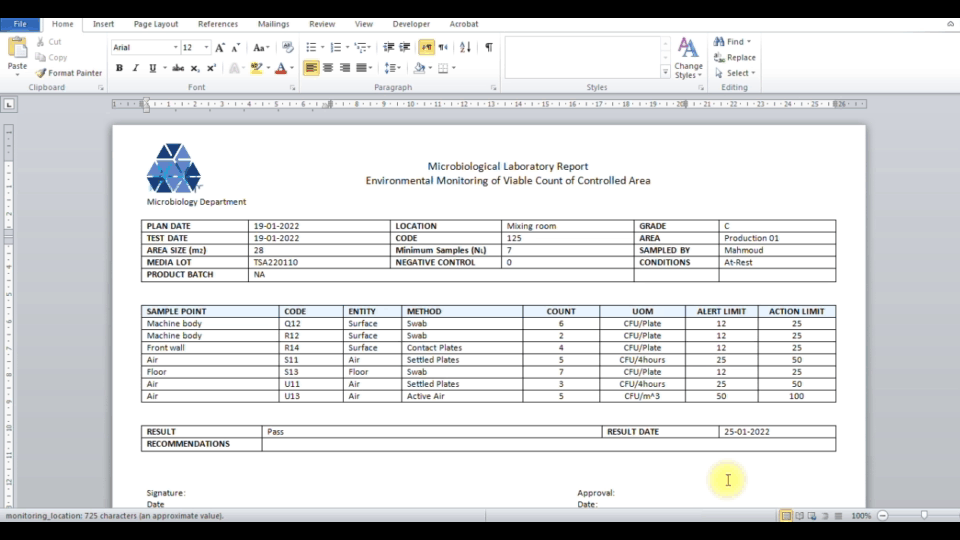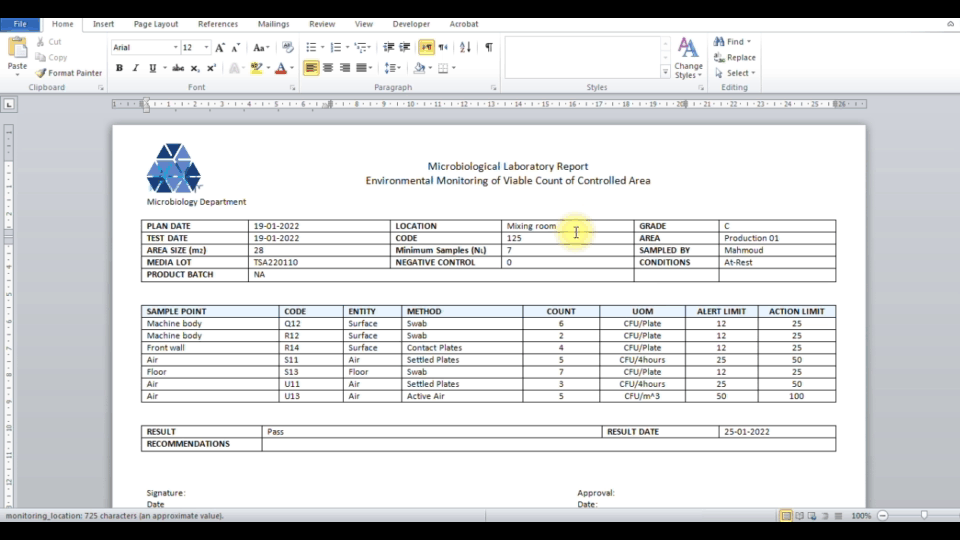
Secure web portal provides 24/7 access to sample status. Download Excel, Word, PDF reports. Samples exceeding limits are automatically highlighted.
Ensuring Pristine Cleanliness
Maintaining immaculate sterility in controlled environments is paramount across various industries, from pharmaceutical manufacturing to food processing. Contamination can compromise product quality and endanger health.
Cleantrol EM offers expertise, validation, and continuous improvement. It uses advanced microbial detection techniques to provide a robust assessment of your critical spaces.
- ✓ Expertise: Guidance on contamination control.
- ✓ Compliance: Adherence to stringent international standards.
- ✓ Peace of Mind: Safeguard products and brand reputation.

Take Control of Your Environment
Contact us to see how Cleantrol EM can secure your critical spaces.
Request Demo


